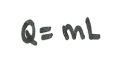A2 Physics
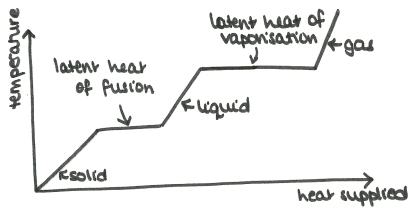
The latent heat, L, of a substance is the energy needed to change the state of a unit mass. This is measured in Jkg-1.
There is no temperature change until the substance has changed state completely.
There are three types of latent heat:
![]() Fusion: between solid and liquid
Fusion: between solid and liquid
![]() Vaporisation: between liquid and gas
Vaporisation: between liquid and gas
![]() Sublimation: between solid and gas
Sublimation: between solid and gas
So when a substance is heated, the temperature changes until it reaches the latent heat, then remains at the same temperature until all of it has changed state: